Chemistry

Chemicals in flasks (including ammonium hydroxide and nitric acid) lit in different colors

Democritus' atomist philosophy was later adopted by Epicurus (341–270 BCE).

Antoine-Laurent de Lavoisier is considered the "Father of Modern Chemistry"

Top: Expected results: alpha particles passing through the plum pudding model of the atom undisturbed.
Bottom: Observed results: a small portion of the particles were deflected, indicating a small, concentrated charge.
Bottom: Observed results: a small portion of the particles were deflected, indicating a small, concentrated charge.

Laboratory, Institute of Biochemistry, University of Cologne.
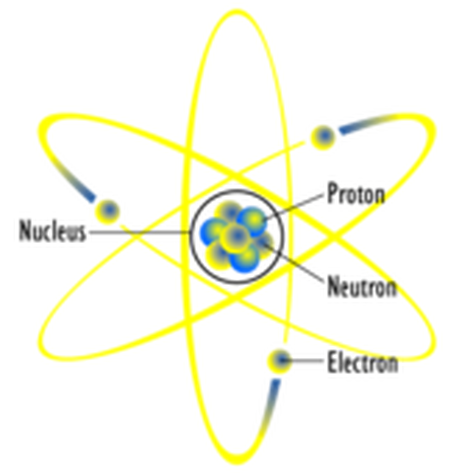
A diagram of an atom based on the Rutherford model
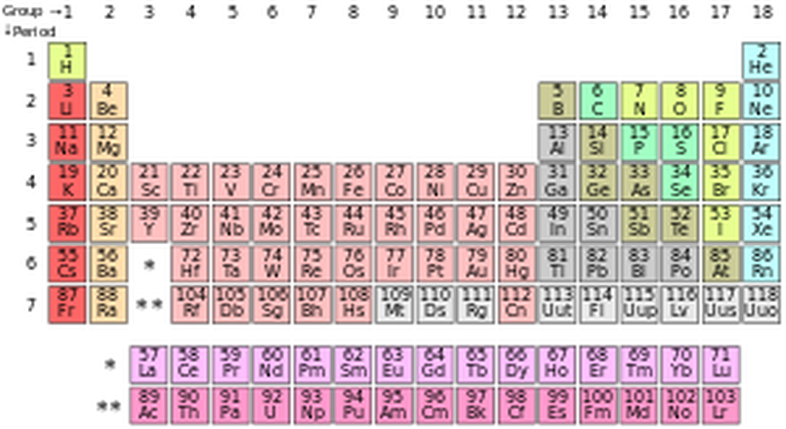
Standard form of the periodic table of chemical elements. The colors represent different categories of elements
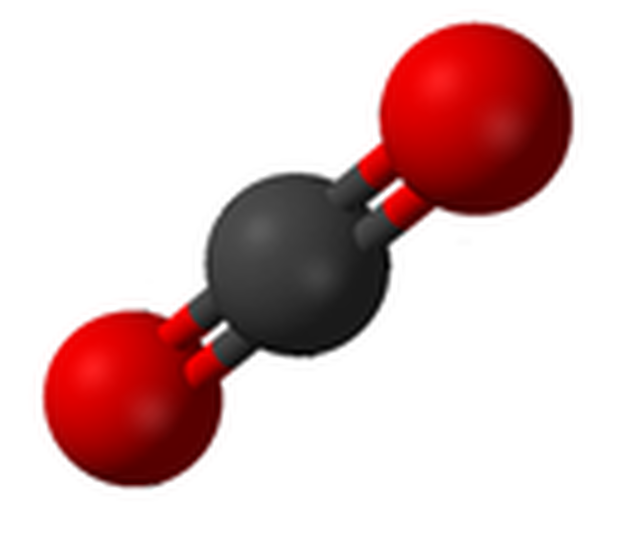
Carbon dioxide (CO2), an example of a chemical compound
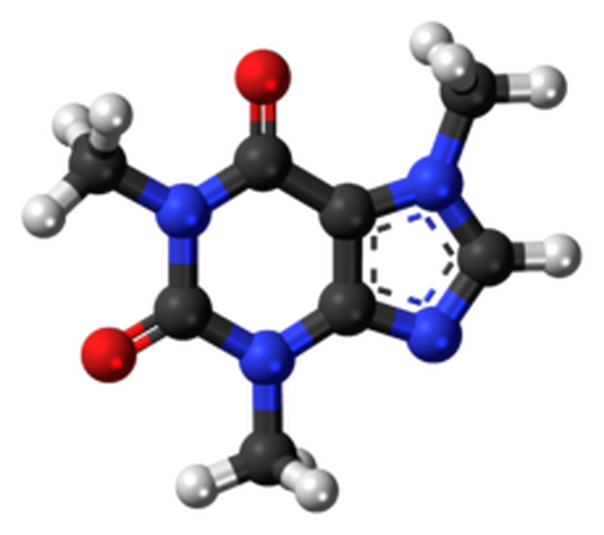
A ball-and-stick representation of thecaffeine molecule (C8H10N4O2).
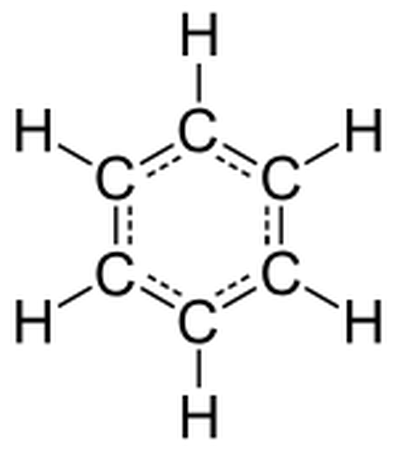
A 2-D skeletal model of abenzene molecule (C6H6)
Examples of pure chemical substances. From left to right: the elements tin (Sn) and sulfur (S),diamond (an allotrope of carbon), sucrose (pure sugar), and sodium chloride (salt) and sodium bicarbonate (baking soda), which are both ionic compounds.
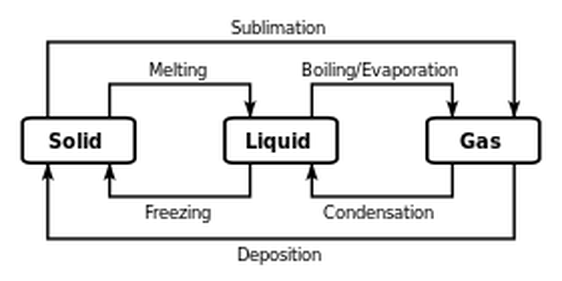
Example of phase changes
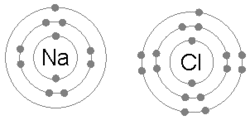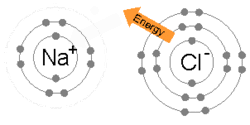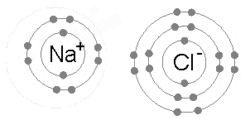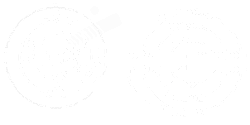
An animation of the process of ionic bonding between sodium (Na) and chlorine (Cl) to formsodium chloride, or common table salt. Ionic bonding involves one atom taking valence electrons from another (as opposed to sharing, which occurs in covalent bonding
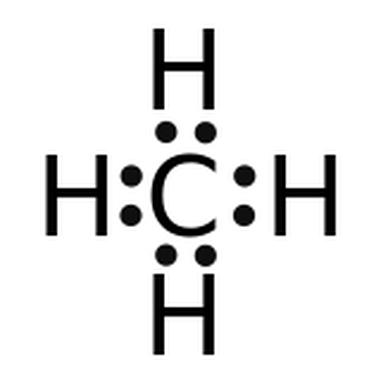
In the methane molecule (CH4), the carbon atom shares a pair of valence electrons with each of the four hydrogen atoms. Thus, the octet rule is satisfied for C-atom (it has eight electrons in its valence shell) and the duet rule is satisfied for the H-atoms (they have two electrons in their valence shells.
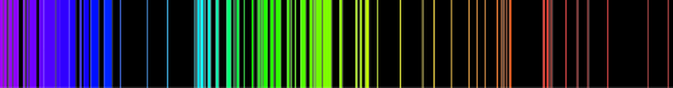
Emission spectrum of iron

During chemical reactions, bonds between atoms break and form, resulting in different substances with different properties. In a blast furnace, iron oxide, acompound, reacts with carbon monoxide to form iron, one of the chemical elements, and carbon dioxide.

The crystal lattice structure of potassium chloride (KCl), a salt which is formed due to the attraction of K+ cations and Cl-anions. Note how the overall charge of the ionic compound is zero.
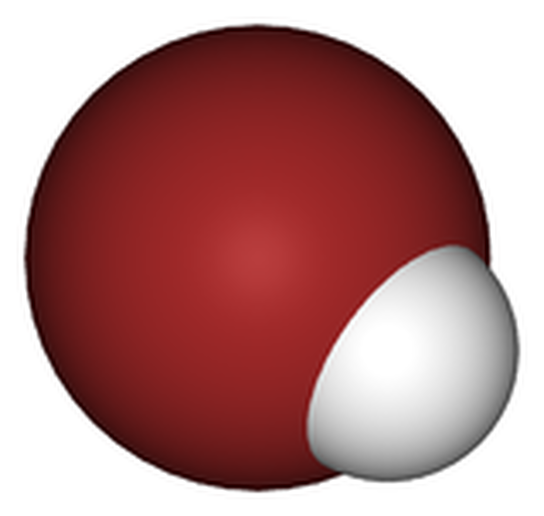
When hydrogen bromide(HBr), pictured, is dissolved in water, it forms the strong acid hydrobromic acid